Engineering Precision in Neuromodulation: Why Goochain is the Trusted Partner for Global taVNS Brands
In the rapidly evolving field of Transcutaneous Auricular Vagus Nerve Stimulation (taVNS), hardware reliability is the bridge between a scientific concept and a successful medical or wellness product. Goochain (Dongguan Goochain Technology Co., Ltd.), as a source manufacturer with deep R&D roots, sets the industry standard for taVNS/tVNS hardware.
Reliability starts with a rigorous quality management system. Goochain operates under the ISO 13485:2016 medical quality standard (Certificate No: 64622B8060236R1S). As a Class I registered production facility, we provide the regulatory security that global brands need to navigate complex compliance landscapes.
The therapeutic efficacy of VNS depends on precise stimulation of the Cymba Concha. Goochain’s patented ear clip electrodes feature flexible conductive silicone. This unique design automatically adjusts to the individual ear size and curvature of each user, ensuring:

Unlike standard TENS devices, our hardware is engineered for the neural system. We support Constant Current (CC) technology, maintaining stable output even as skin impedance fluctuates.
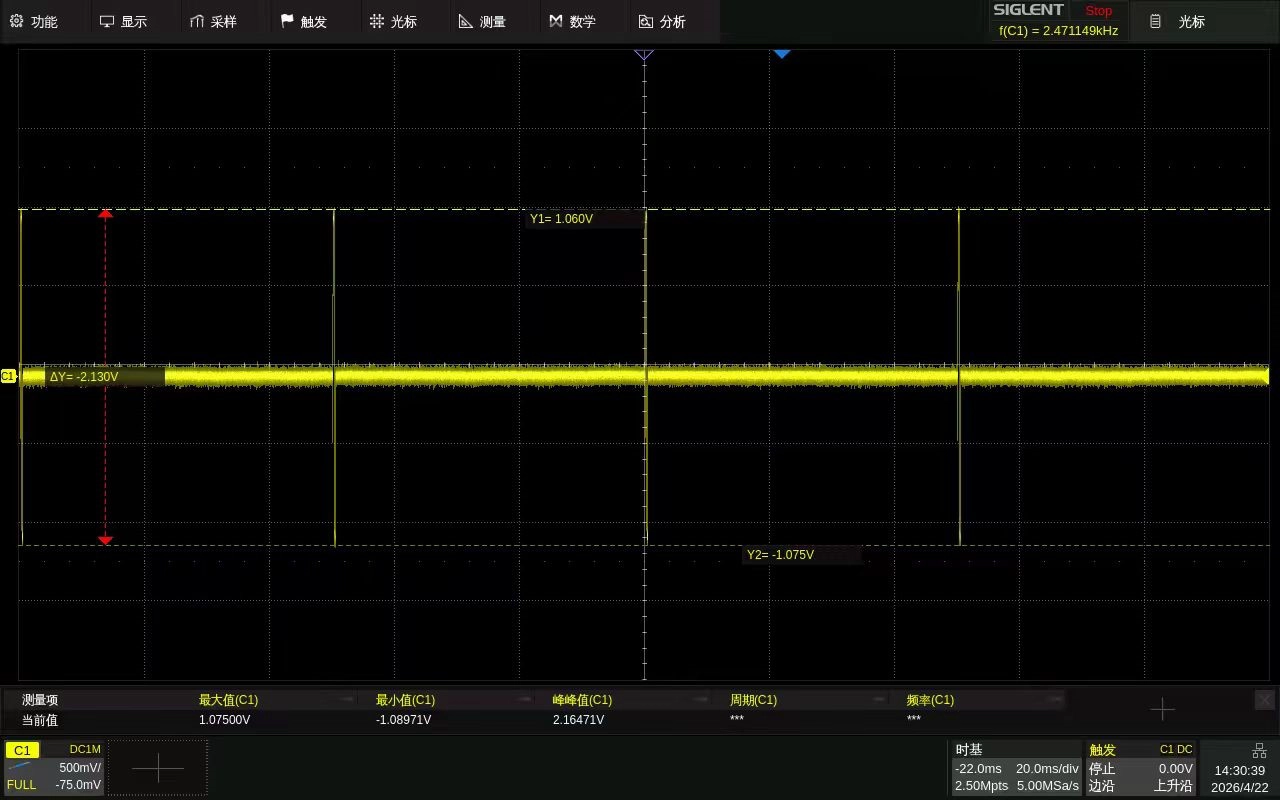
Load: 1 kΩ
Setting: 4
Current: 8 mA / 30 ≈ 0.2666 mA
Current at Setting 4: 4 × 0.2666 mA ≈ 1.0664 mA
Theoretical Voltage: 1.0664 mA × 1 kΩ = 1.0664 V
Actual Measured Voltage: 1.075 V to -1.0897 V
Error:
Positive half-cycle: 0.806%
Negative half-cycle: 2.19%
| Feature | Goochain (Source Factory) | Typical Trading Company |
|---|---|---|
| Quality System | ISO 13485:2016 Certified | No Medical Quality System |
| Regulation | Class I Production Filing | No Manufacturing Record |
| Hardware Control | Constant Current (CC) Technology | Constant Voltage (Unstable) |
| R&D Depth | ID, MD, Electronics, Firmware | Ready-made Samples Only |
| Materials | Medical-grade Conductive Silicone | Unknown/Food-grade Only |
| Durability | 10,000+ Bending Test | No Formal Testing |
| Stage | Deliverables | Timeline |
|---|---|---|
| 1. Concept | Design Brief & Feasibility Study | 3-5 Days |
| 2. R&D | 3D Structural Files & PCB Layout | 2-3 Weeks |
| 3. Prototype | Working Functional Samples | 7-10 Days |
| 4. Tooling | Precision Steel Mold Construction | 25-30 Days |
| 5. Production | Mass Production & Medical-Grade QC | 60-Day Cycle |
Q1: Is Goochain's taVNS hardware compliant for global markets?
A: Yes. We are an ISO 13485:2016 certified facility with Class I production filing. Our products meet CE, FCC, UKCA, and RoHS standards.
Q2: Can you customize pulse sequences for clinical research?
A: Absolutely. Our 15-person R&D team supports Bluetooth/App integration and custom firmware for specialized pulse sequences.
Our hardware development is guided by industry-leading neuroscience research:
Contact our Engineering Team today for your custom taVNS project: https://www.cngoochain.com/contact-us.html